
Since 1 milligram is one-millionth of a kilogram, these measurements could also be expressed as parts per million. Molar Mass of Pure Metals Metal Molar mass (g/mol) Actinium (Ac) 227: Aluminum (Al) 26. Interactive periodic table of elements - your complete guide to the elements including definition, mass & names of each chemical in the periodic table. masspercent computemasspercentage(elementgroup, dictionary) Compute the number of moles of the sample. The molar mass of any element can be determined by finding the atomic mass of the element on the periodic table. Atomic Mass in the Periodic Table of Elements. The following table provides a comprehensive list of molar mass values for pure metals. molarmass computemolarmass(elementgroup, dictionary) Call the computemasspercentage function to compute the mass percentage of the molecule from the compound list. One mole (abbreviated mol) is equal to 6.022×10 23 molecular entities (Avogadro’s number), and each element has a different molar mass depending on the weight of 6.022×10 23 of its atoms (1 mole). Explore how atomic mass changes with atomic number in the periodic table of elements via interactive plots. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. Each element is represented by its atomic number, symbol, name, and abundance in milligrams per kilogram of Earth’s crust. Explore a comprehensive list of molar mass data for pure metals using our sortable table. 1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. 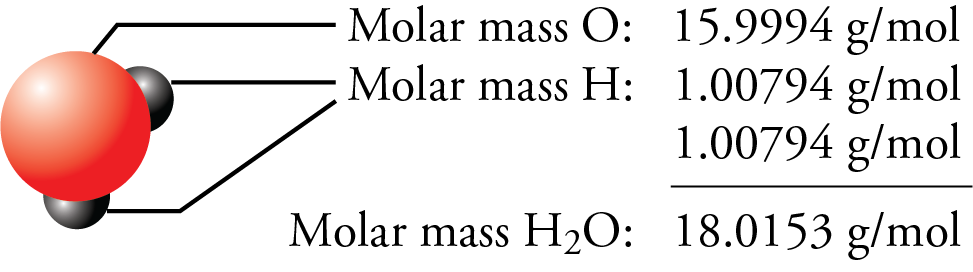
Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
